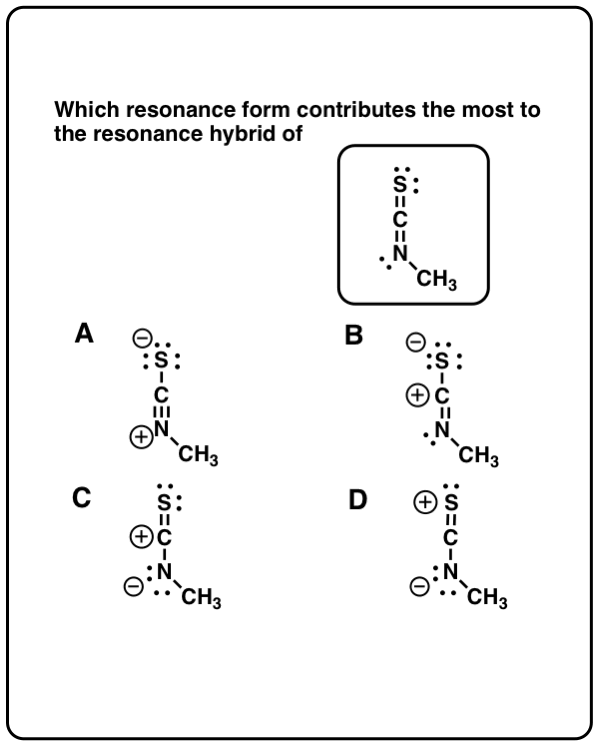
The more the number of resonance structures, more stable is the resonance hybrid.ĥ) When atoms of different electronegativities are involved, the structure with a negative charge on the more electronegative atom and positive charge on less electronegative atom is of lower energy and hence contribute more towards the resonance hybrid than the alternate structure in which the charges are reversed.Ħ) Structure having like charges on adjacent atoms are highly unstable and hence contribute little towards the resonance hybrid.ħ) Structures which help to delocalize the positive charge make important contribution towards the resonance hybrid regardless of the fact whether the positive charge is on less or more electroneagtive atom.Ĩ) Resonance structure in which all the atoms have octet of electrons make larger contribution towards the resonance hybrid the resonance hybrid than those structures in which one or more of the atoms do not have octet of electrons. As far as possible, all the resonance structure should have nearly the same energy.In case of atoms of the second period in the periodic table, such resonance structure which violate octet rule should not be considered.All the resonance structures should have the same number of unpaired electrons. 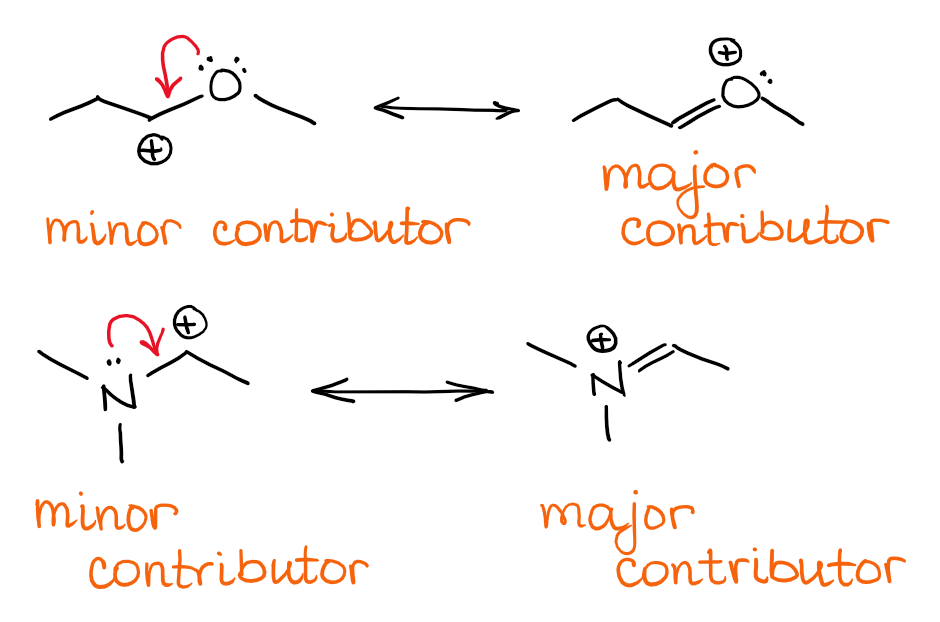
The various resonance structure should differ only in the position of electrons and not in the position of atoms or nuclei.More the number of equivalent resonance structures, greater is the delocalization of electrons, higher is the resonance energy and hence more stable is the compound. It is defined as the difference in internal energy of the resonance hybrid and the most stable canonical structure. The circle inside the ring denotes completely delocalized six π-electrons.Ī resonance hybrid is always more stable than any of its canonical structure.This stability is due to delocalization of electrons and is measured in terms of resonance energy or delocalization energy. 1.39 A° and lie in between carbon-carbon bond length of 1.34 A° and carbon-carbon single bond length of 1.54 A°. The various resonance structure are separated by a double headed arrow (↔)īenzene can be represented as a resonance hybrid of the following two kekule structure :Īny of these Kekule structure, cannot explain all the properties of benzene.According to these structure, a molecule of benzene should have three carbon-carbon single bonds of 1.54 A° length and three carbon-carbon double bonds of 1.34 A° length.But actually it has been found that all the six carbon-carbon bonds in benzene are equal.Īny two adjacent carbon atoms in benzene are neither joined by a pure single bond nor by a pure double bond.As a result, all the carbon-carbon bond lengths are equal i.e. 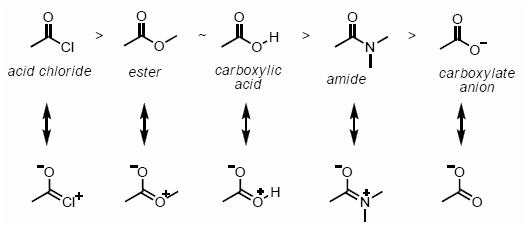
The actual structure of the molecule is not represented by any of the resonance structures but is a resonance hybrid of all these Lewis structure. The various Lewis structure, none of which is capable of describing all the known properties of the compound are called canonical or resonance structures. The phenomenon of resonance is said to occur whenever for a molecule we can write two or more Lewis structure which differ in the position of electrons but not in the relative position of atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
